CELL-MET Researchers Evolve Strategies to Generate Perfusable Vascular Networks
Outcome/Accomplishment
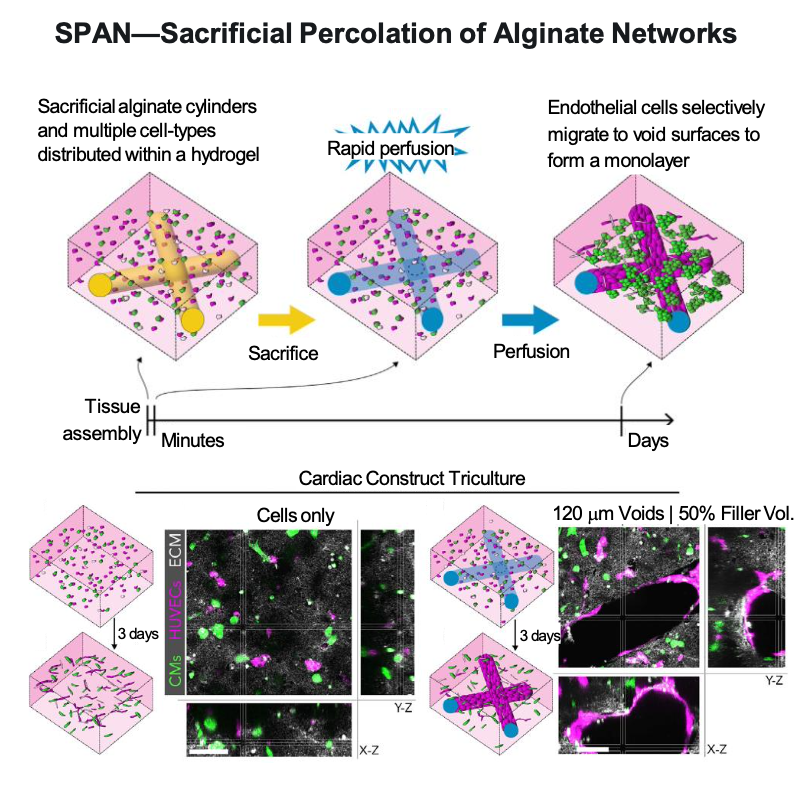
Researchers at the Cellular Metamaterials (CELL-MET) Engineering Research Center (ERC) have developed two different strategies to generate perfusable vascular networks—networks that allow fluids to pump or flow through organs or tissues. In a method called SPAN, a sacrificial network of alginate fibers is integrated within large extracellular matrix (ECM) constructs. Degrading the fibers enables rapid perfusion. The ESCAPE method generates multiscalar vascular networks with high fidelity. CELL-MET is funded by the National Science Foundation (NSF) and based at Boston University, the University of Michigan, and Florida International University.
Impact/Benefits
NSF CELL-MET’s research seeks to generate a centimeter-scale cardiac patch with embedded perfusable vasculature that maintains stable function as measured by tissue viability, structure, and electromechanical function. A key milestone in benchmarking an engineered perfusable vasculature is the demonstration of its ability to deliver nutrients, and most critically oxygen, into a thick engineered tissue that supports survival of cells embedded in the tissue. Both SPAN and ESCAPE can generate vasculature to nourish cardiac tissues. In contrast to the non-vascularized tissue that exhibits significant cell death in inner tissue regions, vascularized tissues will maintain more than 90 percent cell viability. SPAN is particularly applicable to various device and tissue formats and ECMs.
Explanation/Background
Researchers at the Cellular Metamaterials (CELL-MET) Engineering Research Center (ERC) have developed two different strategies to generate perfusable vascular networks—networks that allow fluids to pump or flow through organs or tissues. In a method called SPAN, a sacrificial network of alginate fibers is integrated within large extracellular matrix (ECM) constructs. Degrading the fibers enables rapid perfusion. The ESCAPE method generates multiscalar vascular networks with high fidelity. CELL-MET is funded by the National Science Foundation (NSF) and based at Boston University, the University of Michigan, and Florida International University.
Impact/benefits: NSF CELL-MET’s research seeks to generate a centimeter-scale cardiac patch with embedded perfusable vasculature that maintains stable function as measured by tissue viability, structure, and electromechanical function. A key milestone in benchmarking an engineered perfusable vasculature is the demonstration of its ability to deliver nutrients, and most critically oxygen, into a thick engineered tissue that supports survival of cells embedded in the tissue. Both SPAN and ESCAPE can generate vasculature to nourish cardiac tissues. In contrast to the non-vascularized tissue that exhibits significant cell death in inner tissue regions, vascularized tissues will maintain more than 90 percent cell viability. SPAN is particularly applicable to various device and tissue formats and ECMs.
Explanation/ background: v
NSF CELL-MET’s research team included Christopher S Chen, Alice White, Brendon Baker, Alex Lammers, Subramanian Sundaram and Maggie Jewett. Two research papers, in Nature (https://www.nature.com/articles/s41586-024-08175-5) and Matter (https://pubmed.ncbi.nlm.nih.gov/39221109/), resulted from the work.
Location
Boston, MassachusettsStart Year
Biotechnology and Healthcare
Lead Institution
Core Partners
Fact Sheet
Outcome/Accomplishment
Researchers at the Cellular Metamaterials (CELL-MET) Engineering Research Center (ERC) have developed two different strategies to generate perfusable vascular networks—networks that allow fluids to pump or flow through organs or tissues. In a method called SPAN, a sacrificial network of alginate fibers is integrated within large extracellular matrix (ECM) constructs. Degrading the fibers enables rapid perfusion. The ESCAPE method generates multiscalar vascular networks with high fidelity. CELL-MET is funded by the National Science Foundation (NSF) and based at Boston University, the University of Michigan, and Florida International University.
Location
Boston, MassachusettsStart Year
Biotechnology and Healthcare
Lead Institution
Core Partners
Fact Sheet
Impact/benefits
NSF CELL-MET’s research seeks to generate a centimeter-scale cardiac patch with embedded perfusable vasculature that maintains stable function as measured by tissue viability, structure, and electromechanical function. A key milestone in benchmarking an engineered perfusable vasculature is the demonstration of its ability to deliver nutrients, and most critically oxygen, into a thick engineered tissue that supports survival of cells embedded in the tissue. Both SPAN and ESCAPE can generate vasculature to nourish cardiac tissues. In contrast to the non-vascularized tissue that exhibits significant cell death in inner tissue regions, vascularized tissues will maintain more than 90 percent cell viability. SPAN is particularly applicable to various device and tissue formats and ECMs.
Explanation/Background
Researchers at the Cellular Metamaterials (CELL-MET) Engineering Research Center (ERC) have developed two different strategies to generate perfusable vascular networks—networks that allow fluids to pump or flow through organs or tissues. In a method called SPAN, a sacrificial network of alginate fibers is integrated within large extracellular matrix (ECM) constructs. Degrading the fibers enables rapid perfusion. The ESCAPE method generates multiscalar vascular networks with high fidelity. CELL-MET is funded by the National Science Foundation (NSF) and based at Boston University, the University of Michigan, and Florida International University.
Impact/benefits: NSF CELL-MET’s research seeks to generate a centimeter-scale cardiac patch with embedded perfusable vasculature that maintains stable function as measured by tissue viability, structure, and electromechanical function. A key milestone in benchmarking an engineered perfusable vasculature is the demonstration of its ability to deliver nutrients, and most critically oxygen, into a thick engineered tissue that supports survival of cells embedded in the tissue. Both SPAN and ESCAPE can generate vasculature to nourish cardiac tissues. In contrast to the non-vascularized tissue that exhibits significant cell death in inner tissue regions, vascularized tissues will maintain more than 90 percent cell viability. SPAN is particularly applicable to various device and tissue formats and ECMs.
Explanation/ background: v
NSF CELL-MET’s research team included Christopher S Chen, Alice White, Brendon Baker, Alex Lammers, Subramanian Sundaram and Maggie Jewett. Two research papers, in Nature (https://www.nature.com/articles/s41586-024-08175-5) and Matter (https://pubmed.ncbi.nlm.nih.gov/39221109/), resulted from the work.