Bioavailability Enhancement Strategies Developed for Poorly Soluble Drugs
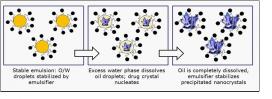
Researchers at the NSF-funded Engineering Research Center (ERC) for Structured Organic Particulate Systems (C-SOPS), headquartered at Rutgers University, have designed a robust emulsion diffusion process for producing nano-suspensions of poorly-soluble weak acid drugs from solvents that are generally recognized as safe (GRAS).
The focus of this work spans the development of effective methods, processes, and control strategies for manufacturing solid dispersions and suspensions containing micro/nano drug particles. The ultimate goal is to increase both the solubility and bioavailability—the active effectiveness—of poorly soluble drugs in flexible drug delivery platforms. Non-destructive and destructive surrogate tests that can help assess the performance of pharmaceutical dosage forms emerge concurrently.
Biopharmaceutical classification system (BCS) Class II drugs are especially characterized by high permeability and low solubility such that their bioavailability is typically limited by their solvation rate. There is generally a correlation between the in vivo (live organic) bioavailability and the in vitro (sample test case) solvation of BCS II drugs. As a result, novel approaches are needed to allow these drugs to release from complex solids into the body more easily. The novel emulsion diffusion process discovered by the C-SOPS team assures that BCS II drugs can overcome both their low oral bioavailability and their erratic absorption rate in the human gastrointestinal (GI) tract, allowing use and dosage of these drugs to become more effective.
The C-SOPS team successfully developed robust nano-emulsion processes that will enhance the active effectiveness of such poorly soluble drugs. Using a trans-oral product across porcine buccal mucosa—an in vitro model widely used to study the permeability of various diffusants and to assess their potential to enter the bloodstream by dissolving in the cheek through the tissues that line the mouth—the researchers achieved immediate release and consequent diffusion of a BCS II poorly soluble drug.
The C-SOPS studies also included nano-suspensions, amorphous processing and solid dispersions. Glassy solid solutions that will increase the solubility of both BCS II and IV drugs were developed, along with dissolution protocols for evaluating how different drugs release from complex solids—such as oral films, solid dispersions, and other dosage forms.