Testing Process Enables Safer Materials for Better Medical Implants
An in vivo biosensor is an implantable device that operates inside the body, such as those for continuous monitoring of glucose, to enable quick and convenient testing at the point of concern. Researchers at the NSF-funded Engineering Research Center (ERC) for Revolutionizing Metallic Biomaterials (RMB) have developed an effective method for testing materials used in biosensors and other biomedical applications to determine important characteristics such as corrosion resistance and biocompatibility. By applying this method they were able to identify carbon nanowire as a safe material for use in the devices. RMB is headquartered at North Carolina A&T State University (NCAT) and has two core partner institutions, the Universities of Pittsburgh and Cincinnati.
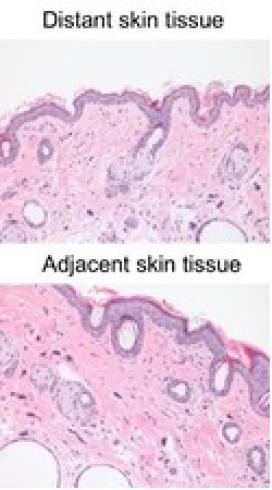
RMB works to develop metallic biomaterials and smart coatings with built-in responsive biosensory capabilities that can adapt to biological changes. This project began with experiments on magnesium (Mg) alloys and single crystal Mg in mice, which would also serve as an animal core for testing biosensors developed by investigators across RMB. Degradation and toxicity of Mg alloy and single crystals implanted under the skin can be easily accessed, monitored, and measured in live animals over a long period of time using an X-ray machine. In combination with data from disease-specific models, information from this study helps to characterize novel materials and develop and modify revolutionary Mg alloys. Carbon nanowire material was tested in mice in RMB’s tenth year. The material did not cause significant discomfort or toxicity to the mice, based on body weight evaluation; did not appear to cause significant pathological alterations to adjacent tissue; and there was no apparent degradation of the material during the experiment period. Applying this new testing approach, RMB was able to show that carbon nanowire could be a safe material for use as wire with a biosensor or other biomedical applications.
For this test, the mouse was anesthetized, then two small incisions were created on two sides of its back and a carbon nanowire was inserted. The two ends of the wire were kept outside of the skin and stabilized with wound clips. The experiment was terminated two weeks later to recover the wire and enable sampling of adjacent and distant site skin for microscope study. The extracted implants were sent to other Center investigators for laboratory analyses, including surface depositions and strength. Host responses to the materials were determined by examining the sections of tissue adjacent to the implants. Potential toxicity of the corroded materials to major organs (heart, lung, brain, liver, kidney, and spleen) was also examined in the tissue as well as blood samples. The expression of inflammatory cytokine genes, which direct the signaling molecule that is secreted from immune cells and certain other cell types that promotes inflammation, as well as the inducible nitric oxide synthase, another important signaling molecule, were quantitatively determined using reverse-transcription polymerase chain reaction to measure the amount of a specific RNA. Deposition and accumulation of Mg in specific organs was measured using inductively coupled plasma mass spectrometry, which atomizes the sample and creates atomic and small polyatomic ions, which are then detected.